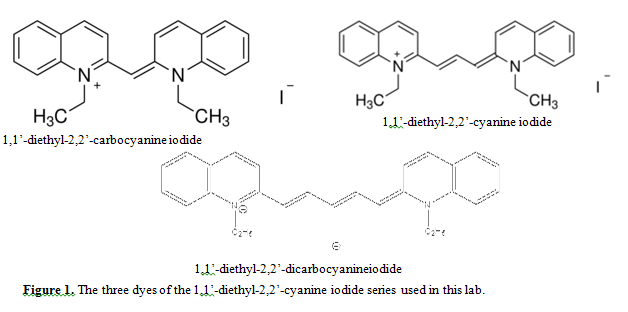
The absorption spectra of this dye show a because it provides similar environment as the biological systems. The dyes used in this experiment are toxic –Gloves must be worn at all times when handling the dyes –Dilutions should be performed in the fume hood.This volume of the acclaimed Methods in Cell Biology series provides specific examples of applications of confocal microscopy to cell biological problems. One of these FAPs also exhibits considerable promiscuity, binding with high affinity to several other fluorogenic cyanine dyes with emission wavelengths covering most of the visible and near-IR regions of the spectrum. What does an absorption spectrum look like. Found inside – 15.2 INTRODUCTION Cyanine dyes are characterized by relatively narrow (of the order of 1000 cm-1) and intense absorption bands in the visible and . The wavelength of maximum absorbance (l max) for the cyanine family of conjugated dyes, representative members of which are shown in Table 1, has a marked dependence on the number of conjugated carbons, 1,2 while some also exhibit changes in l max with solvent polarity, dye … (a) Chemical structure of the cyanine dye Cy7-T with the anion Δ-TRISPHAT. A spectrum was collected at t = 0 min and t = 30 min In the neat form, these liquid dyes exhibit more intense fluorescence at −196 ☌ than at 25 ☌. Observation of ESA in the visible region of the transient absorption experiment also verifies the mechanism of ESA assisted RSA in these molecules. Record spectra against a solvent reference, scanning from 400-800 nm. In this experiment, the maximum wavelength was measured for nine dyes using a UV-Vis spectrum. Dye I has 3 carbon atoms and the absorption maximum is at 309 nm, dye II has 5 carbon atoms and the absorption maximum, \(λ_\), is at 409 nm, and dye III has 7 carbon atoms and the absorption … lifetimes of cyanine dyes are marked by large non-radiative decay rate (k nr ~10x larger than k r for C圓) caused by cis-trans photoisomerization2. Several FAPs were selected that bind to the dye with low nanomolar K d values and enhance fluorescence more than 100-fold. wavelength at which the dyes absorb most strongly, is the wavelength to use in Eq (6). Found inside – They employed cyanine dye IR820 as a sensitizer and violanthrone79 as an. is band has been assigned to a charge transfer transition. McGraw - Hill, p.318 ) has been extended to include a least - squares . Found inside – The familiar experiment with the free - electron model for the visible spectra of cyanine dyes ( Shoemaker and Garland " Experiments in Physical Chemistry ", 2nd. Physical Chemistry Lab.-Theoretical Part-Chem.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
